Now all that's left is a .10$ stock... what has happened? Nothing!
Nasrat's tenure has turned sour, and there has been no news, nothing! But don't take my word for it, listen to the posters on the Ihub board:
There are folks who are looking at the prospects of a better future. One where going sub-zero will be avoided... look at this impressive list. Nasrat, hopefully, is listening...
Nasrat's tenure has turned sour, and there has been no news, nothing! But don't take my word for it, listen to the posters on the Ihub board:
| jour_trader | Sunday, 09/17/17 02:59:39 PM | |
| Re: Couch post# 271030 | ||
| Post # of 271088 | ||
Well, the PPS has gone nowhere but down recently. I wonder why?!
Couch  | Sunday, 09/17/17 03:08:35 PM | |
| Re: jour_trader post# 271035 | ||
| Post # of 271091 | ||
Gee again the PPS was .068 when Nasrat became CEO - the stock is highly undervalued at this point in time on the generics pipeline alone.
There are folks who are looking at the prospects of a better future. One where going sub-zero will be avoided... look at this impressive list. Nasrat, hopefully, is listening...
NASDAQ2020 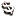 | Sunday, 09/17/17 04:12:32 PM | |
| Re: None | ||
| Post # of 271093 | ||
Elite's Multi-Billion Dollar Platform:
Short to mid term products are entering an IMS market value
of $6 + Billion Dollar$
Bright future ahead for ELTP
Elite's Current Marketed Products and Pipeline is growing rapidly ! Fully developed its worth Billion$
Start watching for the undisclosed drug filings.
$$$$$$$$$$$$
Elite working on 7 AD opiods concurrently
$Generic Products$$
Commercial Product
Branded Product Equivalent
Therapeutic Category
1. Hydromorphone Hydrochloride
8 mg Tablets
Dilaudid®
Pain
2. Naltrexone Hydrochloride
50 mg Tablets
Revia®
Opioid Antagonist
3. Phentermine HCl
37.5 mg Tablets
Adipex-P®
Bariatric
4. Phentermine HCI
15 mg and 30 mg Capsules
Adipex-P®
Bariatric
5. Phendimetrazine Tartrate
35 mg Tablets, USP
Bontril®
Bariatric
6. Isradipine
2.5 mg and 5 mg Capsules
N/A
Cardiovascular
7. Hydroxyzine HCI
10 mg, 25 mg, 50 mg Tablets
Atarax®, Vistaril®
Antihistamine
8. Dantrolene Sodium
25 mg, 50 mg and 100 mg Capsules *
Dantrium®
Skeletal Muscle Relaxant
9. Loxapine
5 mg, 10 mg, 25 mg, 50 mg Capsules *
N/A
Antipsychotic
10. Additional Approved ANDA's *
Undisclosed
Undisclosed
* pending manufacturing site transfer
11. Trimipramine
$BRANDED PRODUCTS$$
Commercial Product
Therapeutic Category
Partner
12. Lodrane D®
OTC Allergy
Valeant Pharmaceuticals International, Inc.
$CONTRACT MANUFACTURING$$
Commercial Product
Branded Product Equivalent
Therapeutic Category
Partner
13. Methadone Hydrochloride
10 mg Tablets
Dolophine®
Pain
Ascend Laboratories, LLC
$GENERIC PIPELINE$$
Programs Under Development
Branded Product Equivalent
Therapeutic Category
Development Stage
14. ELI-300 Opioid
Undisclosed
Pain
Early Development
15. ELI-301 Opioid
Undisclosed
Pain
Early Development
16. Oxycodone HCl and Acetaminophen, USP 325 mg
5 mg, 7.5 mg, 10 mg Tablets
Percocet
Pain
Pending FDA Approval
17. Hydrocodone bitartrate and Acetaminophine tablets, 2.5mg/325mg, 5mg/325mg, 7.5mg/325mg and 10mg/325mg tablets
Norco
Pain
Pending FDA Approval
18. Elite/SunGen Generic #1
Undisclosed
CNS Stimulant
In Development
19. Elite/SunGen Generic #2
Undisclosed
CNS Stimulant
In Development
20. Elite/SunGen Generic #3
Undisclosed
Beta-Blocker
In Development
21. Elite/SunGen Generic #4
Undisclosed
Beta-Blocker
In Development
22. Elite/SunGen Generic #5
Undisclosed
Anti-depressant
In Development
23. Elite/SunGen Generic #6
Undisclosed
Antibiotic #1
In Development
24. Elite/SunGen Generic #7
Undisclosed
Antibiotic #2
In Development
25. Elite/SunGen Generic #8
Undisclosed
Antispasmodic
In Development
26. Generic Troxyca
Pain
27. Generic Embeda
Pain
28. Generic OxyContin
29. Generic Opana Oxymorphone
Pain
$BRANDED PIPELINE$$
Development Stage
Partner
30. $equestOx™ (ELI-200) Opioid
Pain
NDA Submitted
PDUFA date: July 14, 2016
Epic Pharma LLC
31. ELI-201 ER Opioid
Troxyca equivalent
Pain
Pivotal Bio-equivalence
32. ELI-202 Opioid
Pain
Pivotal Bio-equivalence
33. ELI-216 Opioid ER 24hr (once/day)
Pain
Process Development
34. ELI-400 Opioid
Pain
Process Development
35. ELI-500 Opioid
Pain
Process Development
36. ELI-501 Opioid
Pain
Early Development
37. ELI-600 Opioid
Pain
Process development ELTP
38. Chinese ADF opiod
39. Canadian ADF opiod
40. European ADF opiod
$ELTP
ELTP
Short to mid term products are entering an IMS market value
of $6 + Billion Dollar$
Bright future ahead for ELTP
Elite's Current Marketed Products and Pipeline is growing rapidly ! Fully developed its worth Billion$
Start watching for the undisclosed drug filings.
$$$$$$$$$$$$
Elite working on 7 AD opiods concurrently
$Generic Products$$
Commercial Product
Branded Product Equivalent
Therapeutic Category
1. Hydromorphone Hydrochloride
8 mg Tablets
Dilaudid®
Pain
2. Naltrexone Hydrochloride
50 mg Tablets
Revia®
Opioid Antagonist
3. Phentermine HCl
37.5 mg Tablets
Adipex-P®
Bariatric
4. Phentermine HCI
15 mg and 30 mg Capsules
Adipex-P®
Bariatric
5. Phendimetrazine Tartrate
35 mg Tablets, USP
Bontril®
Bariatric
6. Isradipine
2.5 mg and 5 mg Capsules
N/A
Cardiovascular
7. Hydroxyzine HCI
10 mg, 25 mg, 50 mg Tablets
Atarax®, Vistaril®
Antihistamine
8. Dantrolene Sodium
25 mg, 50 mg and 100 mg Capsules *
Dantrium®
Skeletal Muscle Relaxant
9. Loxapine
5 mg, 10 mg, 25 mg, 50 mg Capsules *
N/A
Antipsychotic
10. Additional Approved ANDA's *
Undisclosed
Undisclosed
* pending manufacturing site transfer
11. Trimipramine
$BRANDED PRODUCTS$$
Commercial Product
Therapeutic Category
Partner
12. Lodrane D®
OTC Allergy
Valeant Pharmaceuticals International, Inc.
$CONTRACT MANUFACTURING$$
Commercial Product
Branded Product Equivalent
Therapeutic Category
Partner
13. Methadone Hydrochloride
10 mg Tablets
Dolophine®
Pain
Ascend Laboratories, LLC
$GENERIC PIPELINE$$
Programs Under Development
Branded Product Equivalent
Therapeutic Category
Development Stage
14. ELI-300 Opioid
Undisclosed
Pain
Early Development
15. ELI-301 Opioid
Undisclosed
Pain
Early Development
16. Oxycodone HCl and Acetaminophen, USP 325 mg
5 mg, 7.5 mg, 10 mg Tablets
Percocet
Pain
Pending FDA Approval
17. Hydrocodone bitartrate and Acetaminophine tablets, 2.5mg/325mg, 5mg/325mg, 7.5mg/325mg and 10mg/325mg tablets
Norco
Pain
Pending FDA Approval
18. Elite/SunGen Generic #1
Undisclosed
CNS Stimulant
In Development
19. Elite/SunGen Generic #2
Undisclosed
CNS Stimulant
In Development
20. Elite/SunGen Generic #3
Undisclosed
Beta-Blocker
In Development
21. Elite/SunGen Generic #4
Undisclosed
Beta-Blocker
In Development
22. Elite/SunGen Generic #5
Undisclosed
Anti-depressant
In Development
23. Elite/SunGen Generic #6
Undisclosed
Antibiotic #1
In Development
24. Elite/SunGen Generic #7
Undisclosed
Antibiotic #2
In Development
25. Elite/SunGen Generic #8
Undisclosed
Antispasmodic
In Development
26. Generic Troxyca
Pain
27. Generic Embeda
Pain
28. Generic OxyContin
29. Generic Opana Oxymorphone
Pain
$BRANDED PIPELINE$$
Development Stage
Partner
30. $equestOx™ (ELI-200) Opioid
Pain
NDA Submitted
PDUFA date: July 14, 2016
Epic Pharma LLC
31. ELI-201 ER Opioid
Troxyca equivalent
Pain
Pivotal Bio-equivalence
32. ELI-202 Opioid
Pain
Pivotal Bio-equivalence
33. ELI-216 Opioid ER 24hr (once/day)
Pain
Process Development
34. ELI-400 Opioid
Pain
Process Development
35. ELI-500 Opioid
Pain
Process Development
36. ELI-501 Opioid
Pain
Early Development
37. ELI-600 Opioid
Pain
Process development ELTP
38. Chinese ADF opiod
39. Canadian ADF opiod
40. European ADF opiod
$ELTP
ELTP
No comments:
Post a Comment